|
atomic radius
- one-half of the distance from the center to center of two like atoms Atomic
radius decreases as you go across a period from left to right.
Across the periodic table elements increase in the number of electrons
in their outer energy level and the number of protons in their nucleus.
The addition of positively charged protons and negatively charged electrons
leads to greater attraction and the outer energy level moves closer to
the nucleus.
Atomic radius increases as you go down a group or family. Each row on the periodic table represents an energy level in an atom. As atoms gain energy levels they are added the outter end. In addition, the inner energy level electrons block the attraction between the positively charged nucleus and the electrons in the outer energy level. This shielding effect decreases the attraction between the nucleus and the outer energy level electrons. This allows electrons to drift further away from the nucleus. 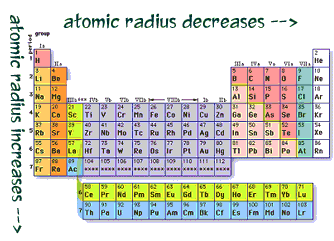 Highest
atomic radius:
cesium(55) : 265
pm
Lowest atomic radius: helium(2) : 31 pm Undefined: (85) (86) (87) (88) (93) (96) (97) (98) (99) (100) (101) (102) (103) (104) (105) (106) (107) (108) (109) (110) (111) (112) (114) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| atomic radius graph X- axis: atomic number Y-axis: atomic radius (pm) 
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
See other element trend graphs: |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||